![]() Acetyl dipeptide-1 cetyl ester
Acetyl dipeptide-1 cetyl ester
Rating : 7
| Evaluation | N. Experts | Evaluation | N. Experts |
|---|---|---|---|
| 1 | 6 | ||
| 2 | 7 | ||
| 3 | 8 | ||
| 4 | 9 | ||
| 5 | 10 |
Pros:
Skin protective (1)10 pts from Al222
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.Evaluate | Where is this found? |
| "Acetyl dipeptide-1 cetyl ester stud" about Acetyl dipeptide-1 cetyl ester Review Consensus 8 by AColumn (9336 pt) | 2022-Jan-24 19:45 |
| Read the full Tiiip | (Send your comment) |
Compendium of the most significant studies with reference to properties, intake, effects.
Ferreira MS, Sousa Lobo JM, Almeida IF. Sensitive skin: Active ingredients on the spotlight. Int J Cosmet Sci. 2021 Nov 23. doi: 10.1111/ics.12754.
Abstract. This study aimed to unveil the most used active ingredients for sensitive skin in facial care products from the pharmacy and parapharmacy channel.
Khmaladze I, Österlund C, Smiljanic S, Hrapovic N, Lafon-Kolb V, Amini N, Xi L, Fabre S. A novel multifunctional skin care formulation with a unique blend of antipollution, brightening and antiaging active complexes. J Cosmet Dermatol. 2020 Jun;19(6):1415-1425. doi: 10.1111/jocd.13176.
Abstract. To develop a unique formula with active complexes, having multifunctional effects for anti-pollution, brightening and anti-aging/barrier strengthening purposes with confirmed activities in vitro and ex vivo skin models, suitable for polluted skin.
Resende DISP, Ferreira MS, Sousa-Lobo JM, Sousa E, Almeida IF. Usage of Synthetic Peptides in Cosmetics for Sensitive Skin. Pharmaceuticals (Basel). 2021 Jul 21;14(8):702. doi: 10.3390/ph14080702.
Abstract. This study aimed to analyze the composition of 88 facial cosmetics for sensitive skin from multinational brands regarding usage of peptides, reviewing their synthetic pathways and the scientific evidence that supports their efficacy.
Sulzberger M, Worthmann AC, Holtzmann U, Buck B, Jung KA, Schoelermann AM, Rippke F, Stäb F, Wenck H, Neufang G, Grönniger E. Effective treatment for sensitive skin: 4-t-butylcyclohexanol and licochalcone A. J Eur Acad Dermatol Venereol. 2016 Feb;30 Suppl 1:9-17. doi: 10.1111/jdv.13529.
Abstract. The objective of this study was to develop an active ingredient concept for the treatment of sensitive skin.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Acetyl dipeptide-1 cetyl ester Review Consensus 10 by Al222 (20707 pt) | 2024-Oct-13 11:40 |
| Read the full Tiiip | (Send your comment) |
Acetyl dipeptide-1 cetyl ester (peptide N-acetyl-L-tyrosyl-L-arginine hexadecyl ester) is a synthetic cosmetic ingredient known for its soothing and anti-inflammatory properties. This biomimetic peptide is designed to mimic the natural functions of the skin, helping to calm irritation and strengthen the skin barrier. It is commonly used in skincare products for sensitive skin, anti-aging creams, and regenerative serums.
Chemical Composition and Structure
Acetyl Dipeptide-1 Cetyl Ester is a dipeptide (composed of two amino acids) with a cetyl ester chain. This structure makes it highly compatible with the skin, allowing it to penetrate effectively and interact with skin receptors to promote a calming and regenerative effect.
The name defines the structure of the molecule
- "Acetyl" indicates the presence of an acetyl group (-COCH3) attached to the molecule. This group is derived from acetic acid.
- "Dipeptide-1" indicates a molecule made up of two amino acids linked together. A dipeptide is a class of peptides, and the "1" might refer to a specific sequence or type of dipeptide.
- "Cetyl ester" indicates the presence of an ester derived from cetyl alcohol. Cetyl alcohol has a chain of 16 carbon atoms, and the ester forms when an acid reacts with cetyl alcohol.
Description of the raw materials used in its production
- Dipeptide-1 - A peptide molecule made up of two amino acids joined by a peptide bond.
- Acetyl Group - An acetyl moiety derived from acetic acid.
- Cetyl Alcohol (or 1-Hexadecanol) - A fatty alcohol used for esterification.
Production Process
Acetyl Dipeptide-1 Cetyl Ester is produced through a chemical synthesis process in which amino acids are combined to form the dipeptide, followed by the addition of a cetyl group to improve lipid and skin compatibility.
- Preparation - Dipeptide-1 is prepared through a peptide synthesis reaction between the desired two amino acids.
- Acetylation - The amino terminal group of dipeptide-1 is acetylated through a reaction with an acetylating agent.
- Esterification - The acetylated dipeptide is subsequently subjected to an esterification reaction with cetyl alcohol to form the cetyl ester.
- Purification - The resulting molecule is purified using techniques such as chromatography to remove impurities and by-products.
Physical Properties
It appears as a white powder or viscous liquid, depending on its form, and is soluble in lipid solvents, insoluble in water and compatible with a wide range of cosmetic formulations. It has high stability and can be incorporated into emulsions, gels, and serums.

Applications
Skincare: Used in creams, lotions, and serums designed for sensitive or reactive skin due to its soothing properties. It is also found in anti-aging products to strengthen the skin barrier and reduce irritation.
Anti-aging Products: Frequently employed in regenerative treatments to improve elasticity and promote skin regeneration.
Functions. Hair conditioner, skin conditioner, anti-wrinkle agent
- Hair conditioning agent. A significant number of ingredients with specific and targeted purposes may co-exist in hair shampoo formulations: cleansers, conditioners, thickeners, matting agents, sequestering agents, fragrances, preservatives, special additives. However, the indispensable ingredients are the cleansers and conditioners as they are necessary and sufficient for hair cleansing and manageability. The others act as commercial and non-essential auxiliaries such as: appearance, fragrance, colouring, etc. Hair conditioning agents have the task of increasing shine, manageability and volume, and reducing static electricity, especially after treatments such as colouring, ironing, waving, drying and brushing. They are, in practice, dispersants that may contain cationic surfactants, thickeners, emollients, polymers. The typology of hair conditioning agents includes: intensive conditioners, instant conditioners, thickening conditioners, drying conditioners. They can perform their task generally accompanied by other different ingredients.
- Skin conditioning agent. It is the mainstay of topical skin treatment as it has the function of restoring, increasing or improving skin tolerance to external factors, including melanocyte tolerance. The most important function of the conditioning agent is to prevent skin dehydration, but the subject is rather complex and involves emollients and humectants that can be added in the formulation.
It is one of the most widely used ingredients in cosmetic products for people who cannot tolerate aggressive ingredients because they have "sensitive skin", "reactive skin" or "intolerant skin" (1).
Acetyl dipeptide-1 cetyl ester relieves the burning sensation and inflammation resulting from exposure of the skin to heat, contact with specific substances such as capsaicin, and mechanical stress (2). In a study comparing the efficacy of two cosmetic products aimed at alleviating skin sensitivity (4-t-butylcyclohexanol in Eucerin® UltraSensitive Soothing Care Dry Skin and Acetyl dipeptide-1 cetyl ester in La Roche-Posay Toleriane® Ultra Intense Soothing Care), both products were found to alleviate the burning sensation, but Acetyl dipeptide-1 cetyl ester demonstrated a faster soothing effect (3).
A viable alternative to Acetyl dipeptide-1 cetyl ester for those who suffer from sensitive skin and have burning or itching sensations on the skin proved, albeit only in in vitro tests, to be the chemical compound 4-t-butylcyclohexanol tested in a 2016 study by M Sulzberger and other private researchers at Beiersdorf in Hamburg, a company that markets cosmetic products including the well-known Nivea (4).
Post-treatment Products: Used in products intended for post-dermatological treatments, such as peels or lasers, to reduce inflammation and support skin repair.
Commercial applications
Soothing Effect. This dipeptide is known for its calming and anti-irritating properties, making it ideal for products meant for sensitive or irritated skin.
Anti-aging Products. Due to its firming and smoothing properties, it can be found in anti-aging creams and serums.
Skin Products. Used in lotions, creams, and serums to enhance skin texture and reduce the feeling of skin tightness.
Soothing Products. Ideal for post-sun exposure or post-hair removal products for its ability to calm and soothe the skin.
Cosmetics. Can be incorporated into cosmetic products to provide additional skin benefits and enhance the overall feel of the product.
Health and Safety Considerations
Safety in Use
Acetyl Dipeptide-1 Cetyl Ester is considered safe for use in cosmetics. No significant adverse reactions have been reported, and it is generally well tolerated by the skin, including sensitive or irritation-prone skin.
Allergic Reactions
Allergic reactions to this peptide are rare, though, as with any cosmetic ingredient, individuals with particularly sensitive skin may experience mild irritation.
Toxicity and Carcinogenicity
It has been extensively evaluated and is deemed safe for use in cosmetic products by major regulatory authorities.
Environmental Considerations
As a synthetic peptide, Acetyl Dipeptide-1 Cetyl Ester is biodegradable and does not pose a significant environmental risk. Its chemical production processes are generally low in environmental impact.
Regulatory Status
Acetyl Dipeptide-1 Cetyl Ester is approved by major regulatory authorities, including the European Union and the Food and Drug Administration (FDA) in the United States, for use in cosmetics.
The most relevant studies on this chemical compound have been selected with a summary of their contents:
Acetyl dipeptide-1 cetyl ester studies
Typical optimal commercial product characteristics Acetyl dipeptide-1 cetyl ester
| Appearance | Fine white powder |
| Density | 1.12±0.1 g/cm3 |
| Purity | 98% |
| Water | <5% |
| Ash | ≤5.0% |
| Loss on Drying | ≤5.0% |
| Heavy metals | NMT 10ppm |
| Arsenic | NMT 2ppm |
| Lead | NMT 2ppm |
| Cadmium | NMT 2ppm |
| Mercury | NMT 2ppm |
| Hygrargyrum | ≤0.1ppm |
| Plate Count | 5,000cfu/g Max |
| Yeast & Mold | 100cfu/g Max |
| PSA | 173.61000 |
| LogP | 8.08350 |
| Water Solubility | Insuluble (6.0E-4 g/L) (25 ºC) |
| Storage | 2~8°C |
 |  |
 |  |
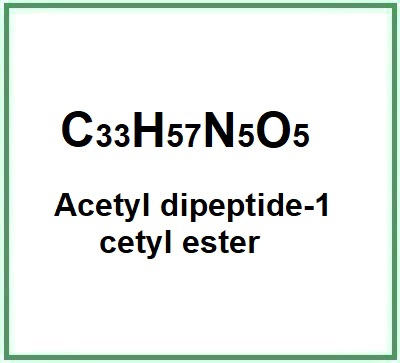
- Molecular Formula C33H57N5O5
- Molecular Weight 603.8
- Exact Mass 603.43600
- CAS 196604-48-5
- UNII 3M7W78X5IR
- EC Number
- DSSTox Substance ID DTXSID00173329
- IUPAC hexadecyl (2S)-2-[[(2S)-2-acetamido-3-(4-hydroxyphenyl)propanoyl]amino]-5-(diaminomethylideneamino)pentanoate
- InChI=1S/C33H57N5O5/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-24-43-32(42)29(18-17-23-36-33(34)35)38-31(41)30(37-26(2)39)25-27-19-21-28(40)22-20-27/h19-22,29-30,40H,3-18,23-25H2,1-2H3,(H,37,39)(H,38,41)(H4,34,35,36)/t29-,30-/m0/s1
- InChl Key JFHZXDZUXGBFAQ-KYJUHHDHSA-N
- SMILES CCCCCCCCCCCCCCCCOC(=O)C(CCCN=C(N)N)NC(=O)C(CC1=CC=C(C=C1)O)NC(=O)C
- MDL number
- PubChem Substance ID
- ChEBI
- ICSC
- NSC
- RTECS
- UN
- NCI
Synonyms:
- acetyl tyrosylarginine cetyl ester
- Hexadecyl (2s)-2-[[(2s)-2-acetamido-3-(4-hydroxyphenyl)propanoyl]amino]-5-(diaminomethylideneamino)pentanoate
- Sensicalmine
References_______________________________________________________________________
(1) Ferreira MS, Sousa Lobo JM, Almeida IF. Sensitive skin: Active ingredients on the spotlight. Int J Cosmet Sci. 2021 Nov 23. doi: 10.1111/ics.12754.
(2) Resende DISP, Ferreira MS, Sousa-Lobo JM, Sousa E, Almeida IF. Usage of Synthetic Peptides in Cosmetics for Sensitive Skin. Pharmaceuticals (Basel). 2021 Jul 21;14(8):702. doi: 10.3390/ph14080702.
Abstract. Sensitive skin is characterized by symptoms of discomfort when exposed to environmental factors. Peptides are used in cosmetics for sensitive skin and stand out as active ingredients for their ability to interact with skin cells by multiple mechanisms, high potency at low dosage and the ability to penetrate the stratum corneum. This study aimed to analyze the composition of 88 facial cosmetics for sensitive skin from multinational brands regarding usage of peptides, reviewing their synthetic pathways and the scientific evidence that supports their efficacy. Peptides were found in 17% of the products analyzed, namely: acetyl dipeptide-1 cetyl ester, palmitoyl tripeptide-8, acetyl tetrapeptide-15, palmitoyl tripeptide-5, acetyl hexapeptide-49, palmitoyl tetrapeptide-7 and palmitoyl oligopeptide. Three out of seven peptides have a neurotransmitter-inhibiting mechanism of action, while another three are signal peptides. Only five peptides present evidence supporting their use in sensitive skin, with only one clinical study including volunteers having this condition. Noteworthy, the available data is mostly found in patents and supplier brochures, and not in randomized placebo-controlled studies. Peptides are useful active ingredients in cosmetics for sensitive skin. Knowing their efficacy and synthetic pathways provides meaningful insight for the development of new and more effective ingredients.
(3) Schoelermann AM, Jung KA, Buck B, Grönniger E, Conzelmann S. Comparison of skin calming effects of cosmetic products containing 4-t-butylcyclohexanol or acetyl dipeptide-1 cetyl ester on capsaicin-induced facial stinging in volunteers with sensitive skin. J Eur Acad Dermatol Venereol. 2016 Feb;30 Suppl 1:18-20. doi: 10.1111/jdv.13530.
(4) Sulzberger M, Worthmann AC, Holtzmann U, Buck B, Jung KA, Schoelermann AM, Rippke F, Stäb F, Wenck H, Neufang G, Grönniger E. Effective treatment for sensitive skin: 4-t-butylcyclohexanol and licochalcone A. J Eur Acad Dermatol Venereol. 2016 Feb;30 Suppl 1:9-17. doi: 10.1111/jdv.13529.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips about this object in __Italiano (2)
Component type: Chemical Main substances: Last update: 2022-01-23 19:56:46 | Chemical Risk: |