![]() E468
E468
Rating : 7
| Evaluation | N. Experts | Evaluation | N. Experts |
|---|---|---|---|
| 1 | 6 | ||
| 2 | 7 | ||
| 3 | 8 | ||
| 4 | 9 | ||
| 5 | 10 |
Cons:
Avoid excessive amounts (1)10 pts from AColumn
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.Evaluate | Where is this found? |
| "Descrizione" about E468 Review Consensus 10 by AColumn (9336 pt) | 2024-Oct-11 18:16 |
| Read the full Tiiip | (Send your comment) |
E468 (Sodium croscarmellose, Croscarmellose Sodium, CNC) is a cross-linked sodium salt of carboxymethyl cellulose, commonly used in pharmaceutical formulations as a superdisintegrant. Its primary function is to facilitate the rapid breakdown of tablets, capsules, or other solid dosage forms in the digestive system, allowing the active ingredients to dissolve more quickly and be absorbed efficiently. It is also used in some cosmetic and personal care products as a thickening or stabilizing agent.
Chemical Composition and Structure
Sodium croscarmellose is derived from cellulose, which is chemically modified by introducing carboxymethyl groups. These groups are then cross-linked to form a network that expands when in contact with water, making it an excellent disintegrant. Its cross-linked structure enhances water absorption, which in turn promotes rapid tablet disintegration.
Physical Properties
Sodium croscarmellose is typically a white to off-white, odorless, and free-flowing powder. It is water-insoluble but swells rapidly upon contact with water, making it ideal for use as a disintegrant in pharmaceutical tablets and capsules. The swelling action helps break down solid formulations into smaller particles for faster dissolution and absorption of active ingredients.
The name describes the structure of the molecule:
- Croscarmellose refers to a carboxymethyl cellulose that has been cross-linked, i.e. its polymer chains are linked to form a three-dimensional network. Carboxymethyl cellulose is a derivative of cellulose, the main structural component of plant cell walls, which has been modified by the addition of carboxymethyl groups (-CH2-COOH).
- sodium refers to the sodium ions (Na+) associated with the carboxymethyl groups of the polymer. When carboxymethylcellulose is cross-linked, the hydrogen ions (H+) in the carboxy groups (-COOH) are replaced by sodium ions, forming the sodium salt of carboxymethylcellulose, or croscarmellose sodium.
The synthesis process generally involves the reaction of cellulose, a natural polymer derived from plant materials, with a cross-linking agent in an alkaline solution. The cross-linking agent, usually sodium monochloroacetate, reacts with cellulose to introduce carboxymethyl groups, creating carboxymethyl cellulose. This is further cross-linked to create sodium croscarmellose, which has the unique property of swelling rapidly when in contact with water.
In practice:
- Step 1: Preparation of raw materials. The raw materials for the synthesis of croscarmellose sodium are typically cellulose and sodium hydroxide.
- Step 2: Carboxymethylation. The cellulose is reacted with sodium hydroxide and chloroacetic acid to introduce carboxymethyl groups on the spine of the cellulose, creating carboxymethylcellulose.
- Step 3: Crosslinking. The carboxymethylcellulose is then crossed to form croscarmellose. This is typically done using a crosslinking agent such as epichlorohydrin.
- Step 4: Purification and drying. The resulting croscarmellose sodium is then purified, typically through filtration and washing, and dried.
It occurs as a fine white, fibrous, odourless powder. It is insoluble in ether, ethanol, acetone or toluene. In contact with water, the volume expands 4 to 8 times its original volume.

What it is used for and where
Medical and Pharmaceuticals
It is an emulsifying disintegrant formed from crude cellulose treated with sodium compounds. It is used to facilitate the breaking up of tablets to improve and accelerate the release of the drug in the intestine after oral administration.
Together with crospovidone, sodium starch glycolate, and others, it is considered a superdisintegrant (1).
Tablets with the same total concentration of superdisintegrant dissolve at a faster rate when the superdisintegrant is included intragranularly (2).
The results of this study demonstrate the low toxicity and safe use of Croscarmellose sodium in oral applications such as pharmaceuticals, dietary supplements, and sweetener tablets (3).
Food
Labelled as E468 in the list of European thickener additives. Food thickeners are normally used to facilitate the ingestion of drugs in tablet form. Some thickeners directly influence the dissolution and disintegration of tablets and may even delay their dissolution.
Excessive intake of celluloses such as E468 may be associated with high risks of cardiovascular diseases (CVD).(4)
Typical optimal characteristics of CROSCARMELLOSE SODIUM as commercial product
| Appearance | White free following powder odorless |
| Grade | Ph.Eur.,JP,NF |
| pH | 5.0~7.0 (10 g/l, H₂O, 20 °C) |
| Density bulk | 0.4 kg/m3 |
| Solubility | 30 g/l |
| Na-chloride&Na-glycolate | ≤0.50% |
| Degree of substitution | 0.60~0.80 |
| Heavy metals | ≤10ppm |
| Water soluble substances | <9 |
| Setting volume | 10~30 |
| Ash | 14.0~26% |
| Storage | +2°C e +30°C. |
CROSCARMELLOSE SODIUM
 |  |
CROSCARMELLOSE
 |  |
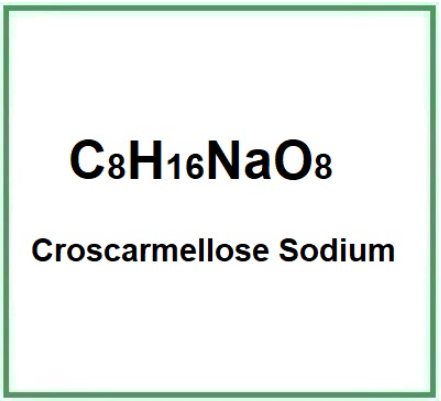
- Molecular Formula C8H16NaO8
- Molecular Weight 263.20
- CAS:74811-65-7 9004-32-4 9000-11-7
- UNII M28OL1HH48
- EC Number: 618-326-2 618-378-6
- DSSTox Substance ID: DTXSID2020555
- InChI=1S/C6H13NO/c1-2-6-5-8-4-3-7-6/h6-7H,2-5H2,1H3/t6-/m0/s1
- SMILES O1C([H])([H])C([H])([H])N([H])[C@@]([H])(C([H])([H])C([H])([H])[H])C1([H])[H]
- MDL number
- PubChem Substance ID
Synonyms:
- Croscarmellose Sodium
- CNC
- Carboxymethylcellulose Sodium
- Carboxymethylcellulose
- Cellulose, Carboxymethyl
- Cross-linked carboxymethylcellulose sodium
References__________________________________________________________________
(1) Yousaf AM, Naheed F, Shahzad Y, Hussain T, Mahmood T. Influence of sodium starch glycolate, croscarmellose sodium and crospovidone on disintegration and dissolution of stevia-loaded tablets. Polim Med. 2019;49(1):19–26. doi:10.17219/pim/111516
Abstract. Background: Sugar substitutes are used by diabetic, obese and calorie-conscious people. As artificial sweeteners are harmful to the body, natural sweeteners are more suitable. Sugar substitutes are available on the market in tablet forms, which are added to hot or cold drinks. Rapid disintegration and dissolution of sugar substitute-loaded tablet is desired. However, the tablets should be hard enough to maintain their integrity during mechanical shocks. Objectives: The objective of this research was to develop rapidly disintegrating and dissolving stevia-loaded tablets with appropriate wetting, hardness and friability....Conclusions: The tablet consisting of stevia, crospovidone, lactose, and magnesium stearate at the weight ratio of 15/2.5/32/0.5 showed excellent results with regards to dissolution and disintegration; accordingly, this formulation could be a potential sugar substitute for diabetic, obese and/or calorie-conscious individuals.
(2) Gordon MS, Chatterjee B, Chowhan ZT. Effect of the mode of croscarmellose sodium incorporation on tablet dissolution and friability. J Pharm Sci. 1990;79(1):43–47. doi:10.1002/jps.2600790111
Abstract. A computer-optimized experimental design was used to study the effect of incorporating a "super disintegrant", croscarmellose sodium, intragranularly, extragranularly, or distributed equally between the two phases of a tablet in which a poorly soluble drug constituted at least 92.5% of the formulation. The results were analyzed by means of a general quadratic response surface model and suggest that tablets with the same total concentration of super disintegrant dissolve at a faster rate when the super disintegrant is included intragranularly. Tablet friability was not affected by the method of super disintegrant incorporation.
(3) Freeman C, Weiner ML, Kotkoskie LA, Borzelleca J, Butt M. Subchronic and developmental toxicity studies in rats with Ac-Di-Sol croscarmellose sodium. Int J Toxicol. 2003;22(3):149–157. doi:10.1080/10915810305108
Abstract. Studies were conducted to evaluate the subchronic and developmental toxicity of Ac-Di-Sol (croscarmellose sodium). In the subchronic study, groups of Sprague-Dawley rats (20/sex/group) received 0 (control), 10000, or 50000 ppm Ac-Di-Sol in the diet for 90 consecutive days (equivalent to 757 and 893 mg/kg/day for males and females fed 10000 ppm, respectively, and to 3922 and 4721 mg/kg/day for males and females fed 50000 ppm, respectively). No mortality, clinical signs of toxicity, or adverse toxicological effects on hematology or serum chemistry parameters, feed consumption, or ophthalmologic examinations were noted in any treatment group. Body weight gain was depressed in high-dose males during the final 3 weeks. The only treatment-related histological lesion noted was moderate renal mineralization at the corticomedullary junction in one high-dose female. This lesion was not considered a specific effect of Ac-Di-Sol, but rather a secondary effect resulting from a potential increase in urinary pH and renal excretion of sodium due to the high intake of sodium associated with Ac-Di-Sol. In the developmental toxicity study, groups of pregnant Sprague-Dawley rats (25/group) received 0 (control), 10000, or 50000 ppm Ac-Di-Sol in the diet on gestational days 6 to 15. No evidence of maternal, fetal, or embryo toxicity was noted. The no-observed-adverse-effect level (NOAEL) for Ac-Di-Sol in both studies exceeds 50000 ppm in the diet, which represents doses of 3922 and 4712 mg/kg/day, for males and females, respectively. The results of these studies demonstrate the low subchronic oral toxicity and developmental toxicity of Ac-Di-Sol, and support the safe use of Ac-Di-Sol in oral applications such as pharmaceuticals, dietary supplements, and sweetener tablets.
(4) Sellem L, Srour B, Javaux G, Chazelas E, Chassaing B, Viennois E, Debras C, Salamé C, Druesne-Pecollo N, Esseddik Y, de Edelenyi FS, Agaësse C, De Sa A, Lutchia R, Louveau E, Huybrechts I, Pierre F, Coumoul X, Fezeu LK, Julia C, Kesse-Guyot E, Allès B, Galan P, Hercberg S, Deschasaux-Tanguy M, Touvier M. Food additive emulsifiers and risk of cardiovascular disease in the NutriNet-Santé cohort: prospective cohort study. BMJ. 2023 Sep 6;382:e076058. doi: 10.1136/bmj-2023-076058. PMID: 37673430; PMCID: PMC10480690.
Arca HC, Mosquera-Giraldo LI, Bi V, Xu D, Taylor LS, Edgar KJ. Pharmaceutical Applications of Cellulose Ethers and Cellulose Ether Esters. Biomacromolecules. 2018 Jul 9;19(7):2351-2376. doi: 10.1021/acs.biomac.8b00517.
Abstract. Cellulose ethers have proven to be highly useful natural-based polymers, finding application in areas including food, personal care products, oil field chemicals, construction, paper, adhesives, and textiles. They have particular value in pharmaceutical applications due to characteristics including high glass transition temperatures, high chemical and photochemical stability, solubility, limited crystallinity, hydrogen bonding capability, and low toxicity. With regard to toxicity, cellulose ethers have essentially no ability to permeate through gastrointestinal enterocytes and many are already in formulations approved by the U.S. Food and Drug Administration. We review pharmaceutical applications of these valuable polymers from a structure-property-function perspective, discussing each important commercial cellulose ether class; carboxymethyl cellulose, methyl cellulose, hydroxypropylcellulose, hydroxypropyl methyl cellulose, and ethyl cellulose, and cellulose ether esters including hydroxypropyl methyl cellulose acetate succinate and carboxymethyl cellulose acetate butyrate. We also summarize their syntheses, basic material properties, and key pharmaceutical applications.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips about this object in __Italiano (1)
Component type: Chemical Main substances: Last update: 2023-04-15 21:10:42 | Chemical Risk: |